|
What is the molarity when water is added to 2 moles of sodium chloride to make 0.5 LL of solution? a) 1 Mī) 2/.5= 4 M Assume the total number of molecules in a sample of liquid is about 3 million trillion. If water is added to 1 mole of sodium chloride in a flask until the volume of the solution is 1 L, what is the molarity of the solution? The component present in the largest amount is the solvent, and the other components are the solutes. In making this solution, should she add the solute to the solvent or the solvent to the solute? 20/10 = 2 gī) the solvent to the solute Which portion of a soap molecule is nonpolar? the tail Distinguish between a solute and a solvent. A B C How much sodium chloride, in grams, is needed to make 15 LL of a solution that has a concentration of 2.4 of sodium chloride per liter of solution? 15*2.4= 36 g A student is told to use 20.0 gg of sodium chloride to make an aqueous solution that has a concentration of 10.0 g/L (grams of sodium chloride per liter of solution).Īssuming that 20.0 gg of sodium chloride has a volume of 7.50 mL, find what a volume of water will she need to make this solution. Which of the following boxes best represents a solution? the middle with both the red dots spread out everywhere and two pink ovals Rank the following solutions in order of increasing concentration: solution A, 0.5 mole of sucrose in 2.0 LL of solution solution B, 1.0 mole of sucrose in 3.0 LL of solution solution C, 1.5 moles of sucrose in 4.0 LL of solution. What is the molar concentration (molarity) of a solution containing 171 grams sucrose in 1.25 L solution? How many moles of sucrose are there in 2.00 L of a 0.500 molar solution? There are 342 grams of sucrose in 1.00 mole of sucrose. A solution of sucrose that has a concentration of 1 mole of sucrose per liter of solution contains 342 grams of sucrose, which equals 6.023××1023 molecules of sucrose. Molarity is defined by the equation, Molarity=number of moles of solute/liters of solution Rank the following solutions in their increasing order of molarity overlapping solutions with equivalent molarity. One of the most commonly used unit of concentration in chemistry is molarity (MM), or molar concentration, which is the number of moles of solute in 1 liter (LL) of solution. Concentration=amount of solute/amount of solution The concentration of a solution can be expressed in many different ways. 
The concentration of a solution is the amount of solute dissolved in a given quantity of solution. 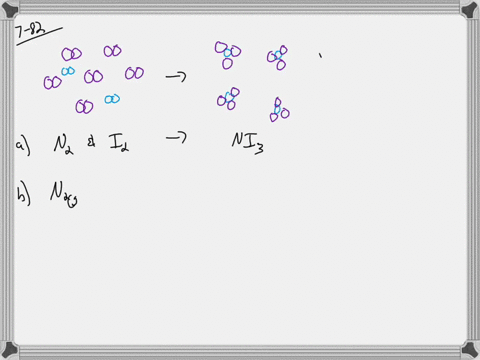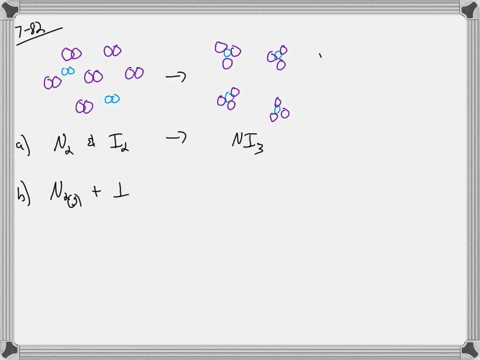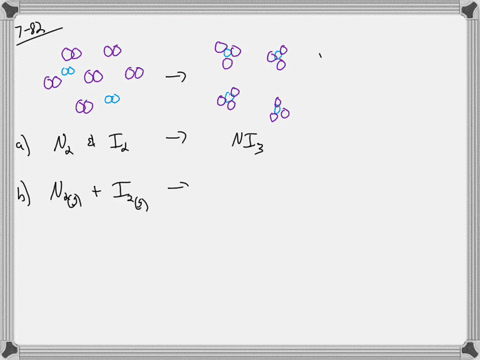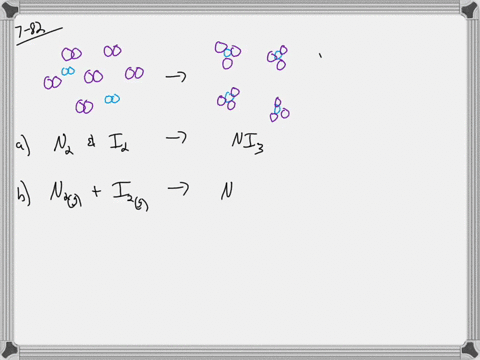
It is always considered as the solvent regardless of amount. In a solution, the component present in a smaller proportion is called the solute while the component present in larger proportion is called the solvent. A solution is a homogeneous mixture of two or more substances.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
